 
They are defined as a set of atoms that share the same number of valence electrons. Periods are the building blocks of all chemical reactions. Periods Groups In chemistry, a period is a line that represents the number of electrons in an atom A group is a set of atoms that share similar properties Periods are the building blocks of all chemical reactionsĪ group is a set of atoms that have similar properties and behave in similar way They are defined as a set of atoms that share the same number of valence electrons.Ĭan be determined by their physical properties, chemical reactivity, or even their appearance Definitions: What are Periods? All carbon atoms are also electrically neutral, meaning they have no net charge.īut what are periods and groups in chemistry? What are the differences between them? To answer these questions, this article will explain the definitions of periods and groups, as well as their relationship to each other. All carbon atoms are also electrically neutral, meaning they have no net charge. For example, all carbon atoms have six protons and six neutrons in their nuclei, which means they are all chemically identical. Each element has its own unique set of properties and can be found in different forms.Ī group is a set of atoms that share similar properties. The most common example of this is the periodic table of elements. In chemistry, a period is a line that represents the number of electrons in an atom. We also know that helium has two electrons in its outermost shell and can therefore form compounds with other elements. This means that hydrogen can combine with other elements to form compounds. For example, we know that the element hydrogen has one electron in its outermost shell. 
The periodic table is also important because it shows the relationships between elements. It also tells us how to make new elements. The periodic table is important because it shows the chemical properties of elements. The vertical columns are periods and each period has a group number. The periodic table is arranged in order of increasing atomic mass, from left to right (top to bottom). The first period has one element, the second period has two elements, and so on up to the last period which has 17 elements.ĭifference between a Bobcat and a Mountain Lion The number of elements in each period is called the group number. You will see that there are groups of elements in this row. The next time you take out your periodic table, take a look at the bottom row. 
The main difference between periods and groups is that the period has a fixed number of elements while the group has a variable number of elements. Periods are further divided into periods and sub-periods. The Periodic Table has been divided into periods and groups based on their similarities and differences. The main purpose of this table was to help chemists to understand the behavior of different elements. It was first created by Dmitri Mendeleev in 1869. The periodic table has been used for more than 200 years and it is still being used today. 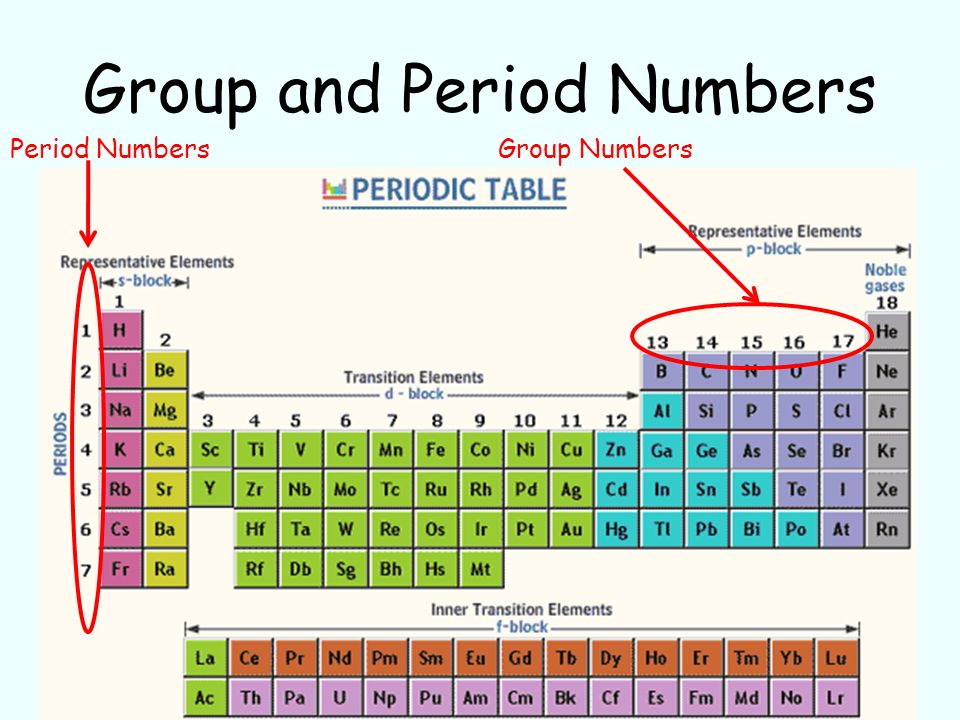
Periodic Table is a table that shows the chemical elements in order of their atomic number. But, you can easily get rid of this confusion by understanding the difference between periods and groups in periodic table. The main reason behind this is that the periodic table has a lot of information in it and it is difficult to comprehend all of them. We all know the feeling of confusion when we try to understand the periodic table. Never disregard professional advice or delay in seeking it because of something you have read on this website! Always seek the advice of your doctor with any questions you may have regarding your medical condition. The Content is not intended to be a substitute for professional medical or legal advice. The contents of the website, such as text, graphics, images, and other material contained on this site (“Content”) are for informational purposes only. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
